Bellinzona, March 4, 2022 – The Bloom’s syndrome helicase (BLM) functions in DNA double-strand break repair to prevent crossover recombination. In the absence of BLM, crossover recombination products are increased, leading to a loss of heterozygosity and dramatically increased incidence of cancers. Mechanistically, BLM helicase functions together with a TopoIIIa topoisomerase to process recombination products to prevent the unwanted outcomes. In a new paper published in Science Advances, the Matos (ETH Zurich, University of Vienna) and the Cejka (IRB Bellinzona) groups now report that the function of BLM is supported by an intricate phosphorylation cascade, including CDK and PLK kinases, as well as the TOPBP1 adaptor protein. A failure to phosphorylate BLM in a timely manner results in the same defects as BLM deficiency. Ilaria Ceppi from IRB Bellinzona, who recently obtained her PhD, is one of the co-first authors of the study.
Article:
Balbo Pogliano, C., I. Ceppi, S. Giovannini, V. Petroulaki, N. Palmer, F. Uliana, M. Gatti, K. Kasaciunaite, R. Freire, R. Seidel, M. Altmeyer, P. Cejka and J. Matos
Sci Adv. 2022; 8:eabk0221
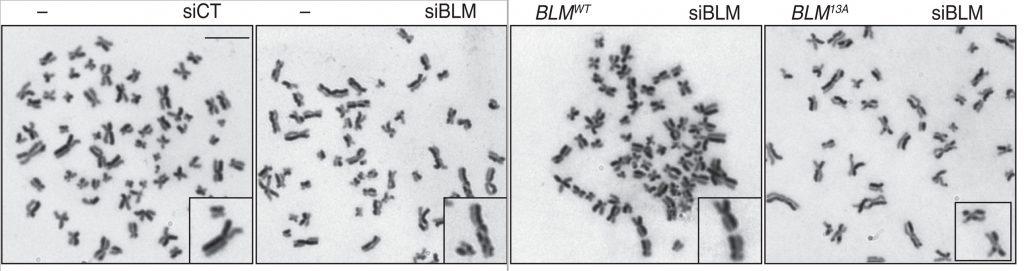
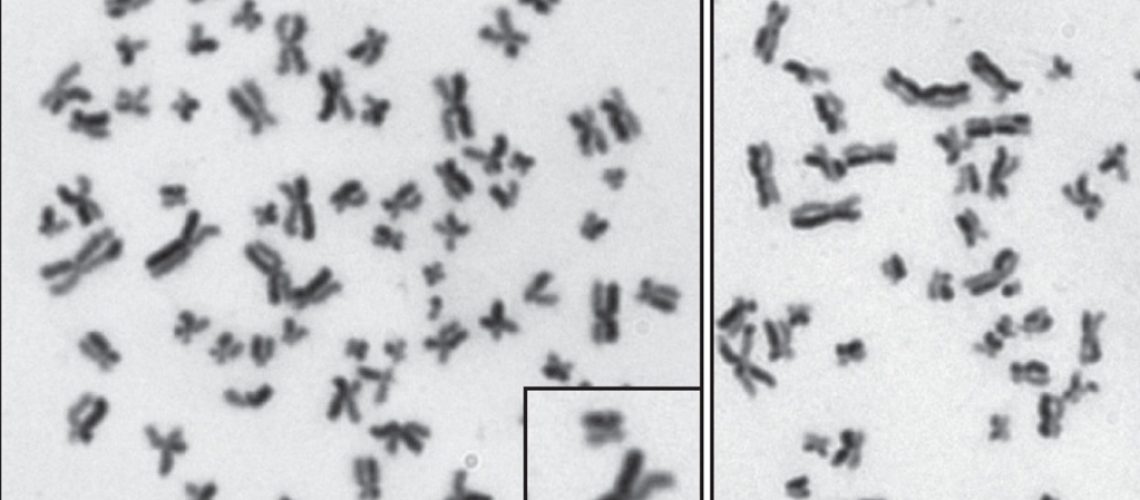
The images indicate sister chromatid exchanges, as readouts for crossover recombination products. These events are rare in BLM-expressing cells, and become much more common in cells lacking BLM, or in cells where BLM cannot be phosphorylated.