After the announcement, in May, of the first results of serological tests carried out on healthcare personnel in Ticino, significant data on the antibody response to SARS-CoV-2 is now available, thanks to the analysis carried out – among others – by the Institute for Research in Biomedicine (IRB, affiliated to USI) and Humabs BioMed (subsidiary of Vir Biotechnology), in close collaboration with hospitals and COVID-19 centers in Ticino (Ente Ospedaliero Cantonale EOC and Clinica Luganese Moncucco). The results of the study were published ‘online first’ in the peer-reviewed journal Cell.
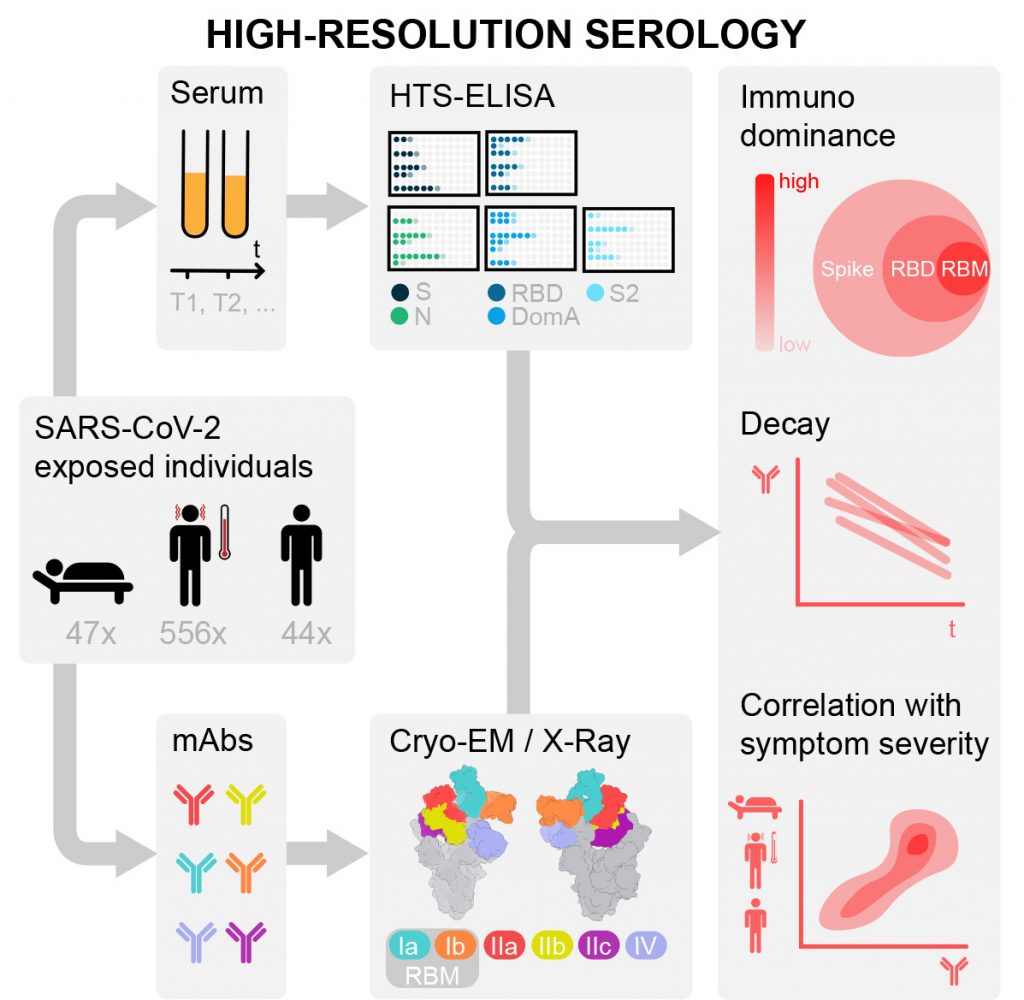
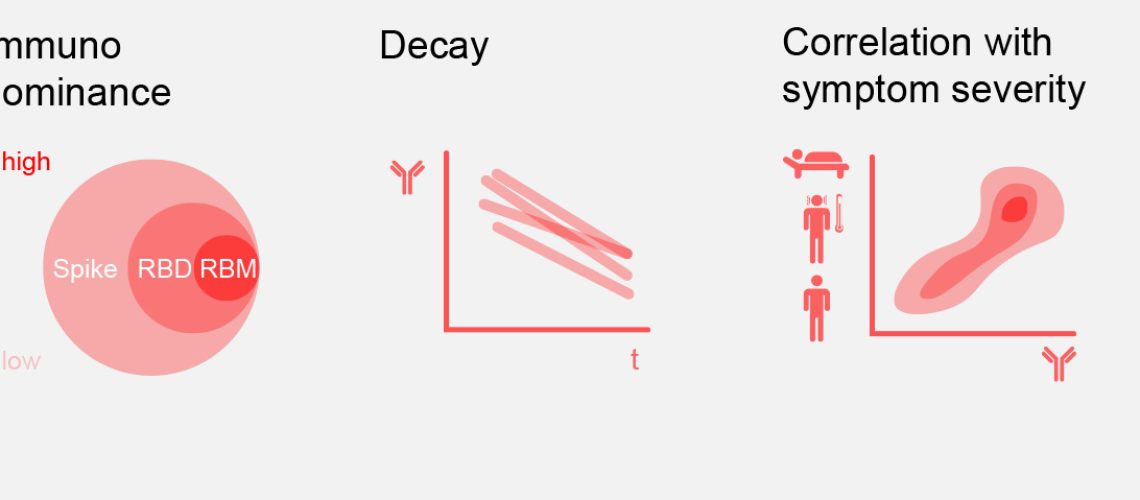
Results of this study, based on blood samples from nearly 650 SARS-CoV-2 infected individuals in Switzerland, Italy and the United States, show that the magnitude of antibodies produced by an infected individual is proportional to disease severity (with hospitalised patients possessing higher antibody titers compared to non-hospitalised patients), and that those antibodies have a half-life of less than two months. The researchers also report that the receptor binding domain (RBD) of the virus is the main target of naturally occurring neutralizing antibodies, accounting for 90% of the neutralizing activity in serum. The study provides new information about the diverse individual antibody response to SARS-CoV-2 infection, and the results establish a blueprint that could help guide future serology studies and inform vaccine and therapeutic design strategies.
The study published on Cell represents a collaborative effort between several hospitals and research institutes in Ticino that have invested significant resources and made significant efforts to ensure the collection, screening and preparation of thousands of samples within a few weeks.
The Chief Medical Officer at EOC, Prof. Dr. med. Paolo Ferrari, says: “Due to the increased risk of exposure of healthcare workers to SARS-CoV-2, in April we started a serum-prevalence study, in collaboration with the IRB, of hospital personnel throughout Ticino. The participation in this study, coordinated by the EOC Clinical Trial Unit (CTU-EOC) under the guidance of Prof. Alessandro Ceschi, was impressive, with over 4,700 samples collected in a few weeks. This work represents an important step forward in the fight against COVID-19, and we are proud of the collaboration with IRB and Humabs BioMed within this project”.
“This study provides new information about the diverse individual antibody response to SARS-CoV-2 infection. The results establish a blueprint that could help guide future serology studies and inform vaccine and therapeutic design strategies,” said Davide Corti, senior vice president of antibody research at Humabs BioMed and study author. “Further, the rapid waning of the natural antibody response and the fact that approximately 60% of infected individuals do not produce antibodies that can block infection underscores the potential need for additional therapeutic approaches.”
Federica Sallusto, Professor at USI and ETH Zurich, and Director of the Center for Medical Immunology at the IRB, which conducted the serological screening of health care workers, comments: “My laboratory was able to quickly refocus its research work on COVID-19 thanks to the collaboration with Humabs BioMed. This work, published in one of the most prestigious scientific journals, is an extraordinary example of how hospitals, research institutes and biotech companies can join forces against the coronavirus”.
Article
Piccoli, L et al.
In Cell, DOI: 10.1016/j.cell.2020.09.037