on July 8, 2019
Our gastrointestinal tract is colonized by bacteria at birth, and, thereafter, a mutualistic interaction with the evolving microbiota is established. The microbiota regulates the metabolic balance of the organism by generating bioactive molecules that are absorbed through the intestinal epithelium. In addition, it stimulates the development of the gut-associated lymphoid tissue (GALT), which comprises the Peyer patches (PPs) in the ileum and generates the adaptive antibody (i.e. secretory Immunoglobulin A, SIgA) response. SIgA protects the organism from invasion by pathogens but also fosters colonization of beneficial microbes.
Adenosine triphosphate (ATP) is an ubiquitous extracellular messenger, which among other ATP responsive P2 receptors, activates the P2X7 subtype in the plasma membrane of T follicular helper (Tfh) cells in the PPs. Tfh cells promote SIgA generation by B cells; P2X7 is stimulated by ATP released by intestinal bacteria and inhibits excessive SIgA responses that could affect microbiota composition. Therefore, extracellular ATP released by the microbiota constitutes an inter-kingdom signalling molecule that regulates the relationship between gut microbes and the local immune system. Because of the lack of this regulatory mechanism, P2X7 deficient mice show enhanced SIgA production and alteration of the microbiota. The group of Fabio Grassi at the IRB-USI of Bellinzona, Switzerland, has previously shown that this altered SIgA response is responsible for metabolic alterations in these mice.
Through the characterization of the faecal microbiota targeted by SIgA in P2X7 deficient mice, the group has now discovered that enhanced SIgA coating of Lactobacillus is responsible for the enrichment of these bacteria and the metabolic alterations observed in these mice. In fact, IRB researchers could reproduce the metabolic alterations by colonizing wild-type mice with Lactobacilli isolated from P2X7 deficient mice. The study, published in Scientific Reports and performed in collaboration with the University of Milan and the University of Bari, Italy, demonstrates that the ATP/P2X7 signaling axis constitutes a crucial regulatory pathway in host/microbiota mutualism to ensure metabolic homeostasis of the organism. Targeting of this axis can be envisaged to condition the extent of colonization of bacteria, like Lactobacilli, which can influence our systemic metabolism.
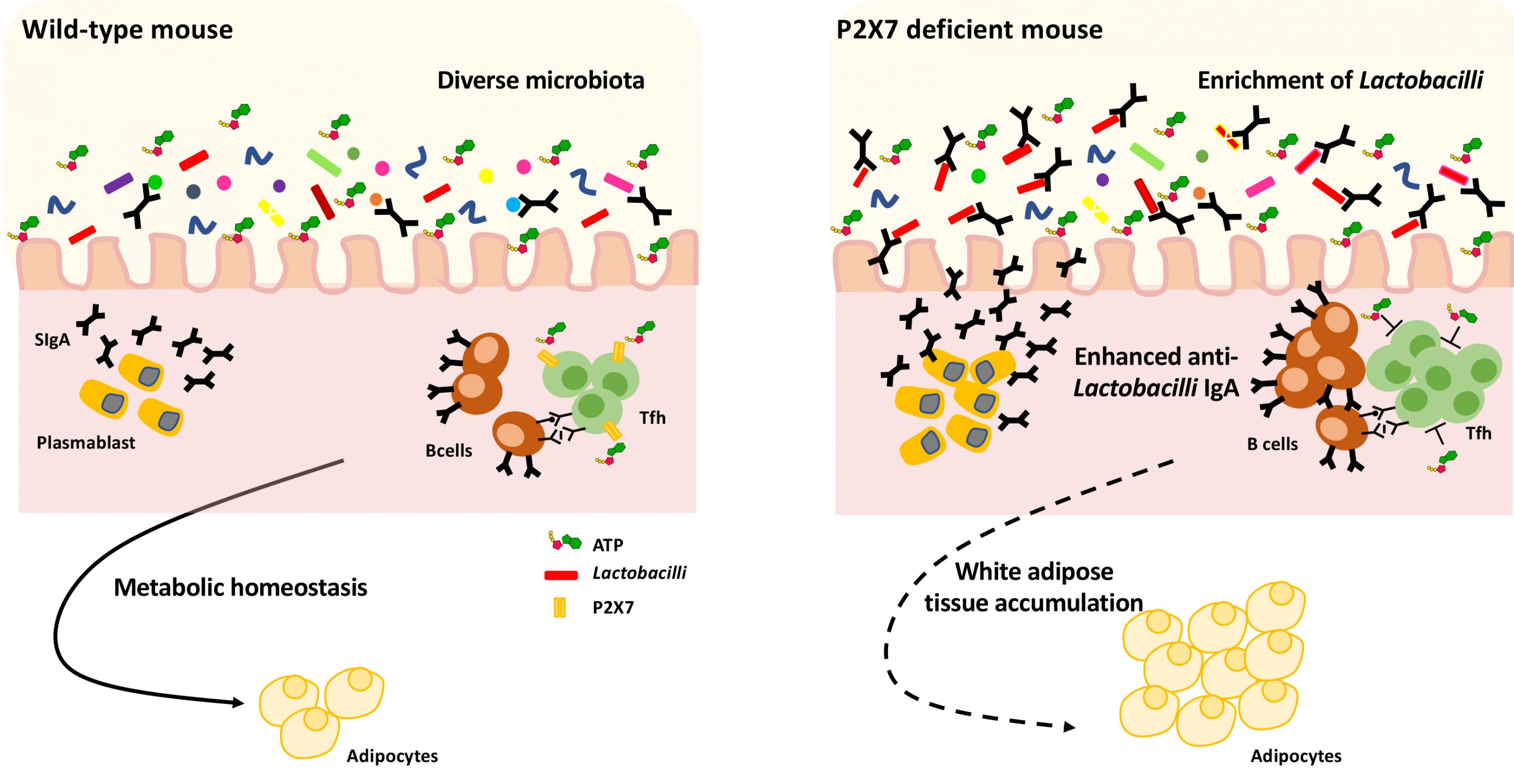 |
| A regulated SIgA response in wild-type mice ensures the maintenance of a beneficial diverse microbiota in the gut (left). The excessive SIgA response in P2X7 deficient mice because of deregulated Tfh cells helper activity and production of SIgA results in enrichment of Lactobacilli and augmented fat deposition (right) |
Article
L. Perruzza, F. Strati, G. Gargari, A. M. D’Erchia, B. Fosso, G. Pesole, S. Guglielmetti, F. Grassi
in Sci Rep (2019) vol. 9 pp9315 , DOI: 10.1038/s41598-019-45724-9