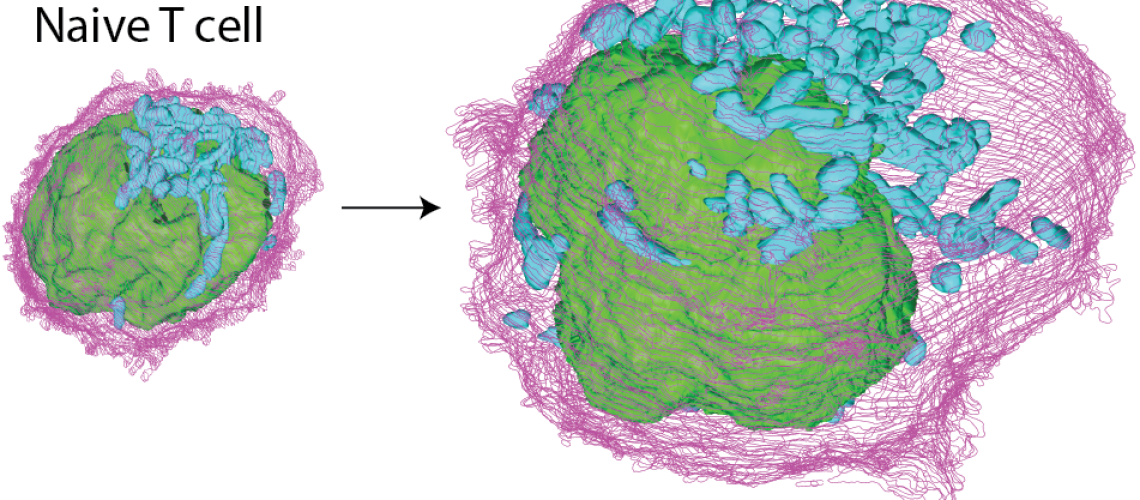
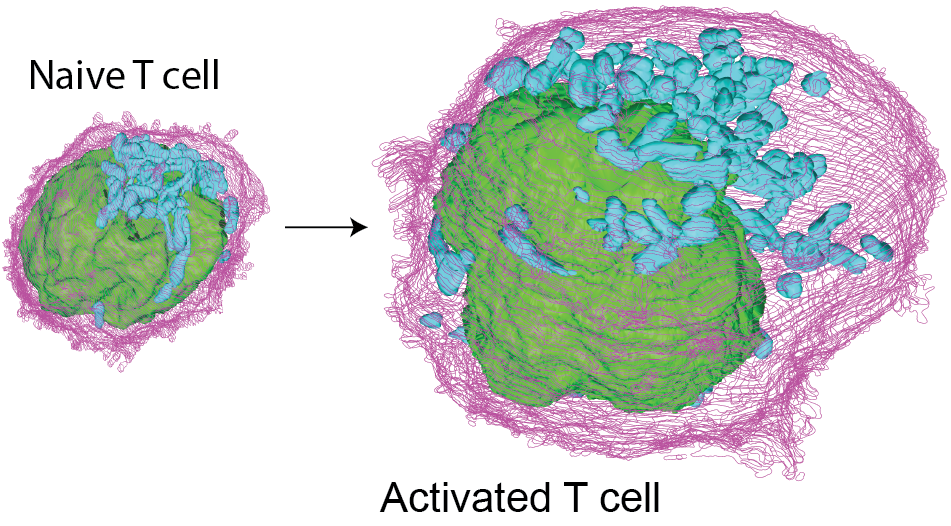
Nearly 200 billion naïve T cells continuously patrol the human body in a quiescent state, prepared to respond to potential threats. Consequently, they may remain inactive for several years in a spore-like state and thus consume little nutrients and energy. However, if they encounter an antigen from a pathogen or a malignant cell, naïve T cells rapidly undergo a remarkable differentiation program to mount a robust immune response to pathogenic threats. Therefore, T cells face a tradeoff between minimizing their metabolic activity while sustaining a maximally prepared state for rapid execution of the activation program.
An international research team led by IRB investigator Roger Geiger used mass spectrometry-based proteomics to analyze the mechanisms that enable quiescent T cells to jump-start their activation program following antigenic recognition. The team found that quiescent, naïve T cells maintain critical proteins and molecular machinery “on standby”, which are only engaged following stimulation. The pre-existing components include idling ribosomes, untranslated mRNAs and an inactive reservoir of glycolytic enzymes. Conversely, T cells also need to downregulate certain components following stimulation, i.e. transcription factors that actively maintain naïve T cells in a quiescent state. The team found that many of the proteins that are down regulated following activation are highly turned over in naïve T cells. When their synthesis stops following stimulation, these proteins are degraded, facilitating their exit from quiescence.
The study also contributes to a quantitative understanding of the T cell response. By implementing a pulsed SILAC approach, absolute protein synthesis rates for thousands of proteins in naïve T cells were determined. In addition, an algorithm to estimate mRNA copy numbers based on RNA-Seq data and mRNA quantifications was developed. Integration of the two datasets allowed the global quantification of mRNA translation kinetics, which is how many times a given mRNA species is read off by ribosomes.
Article
Dynamics in protein translation sustaining T cell preparedness
Tobias Wolf, Wenjie Jin, Giada Zoppi, Ian A. Vogel, Murodzhon Akhmedov, Christopher K. E. Bleck, Tim Beltraminelli, Jan C. Rieckmann, Neftali J. Ramirez, Marco Benevento, Samuele Notarbartolo, Dirk Bumann, Felix Meissner, Bodo Grimbacher, Matthias Mann, Antonio Lanzavecchia, Federica Sallusto, Ivo Kwee & Roger Geiger
Nature Immunology (2020); DOI: 10.1038/s41590-020-0714-5
See also News & Views
This article made the cover of the Nature Immunology Issue.