While populations around the world are being vaccinated against SARS-CoV-2, the U.S. Food and Drug Administration has announced the approval of a treatment against another well-known virus, much less widespread but extremely lethal: Ebola. The new drug was made possible thanks to the fundamental work performed at the laboratories of the Institute for Research in Biomedicine (IRB, affiliated to USI) and Humabs BioMed (subsidiary of the U.S. Vir Biotechnology) in Bellinzona.
The drug is called Ebanga and is produced by Ridgeback Biotherapeutics LP, a U.S. company based in Miami, Florida, specialising in antiviral drugs and therapies. In late December, the FDA announced approval of Ebanga for the treatment of Zaire ebolavirus (Ebolavirus) infection in adults and children. Ebanga blocks binding of the virus to the cell receptor, preventing its entry into the cell.
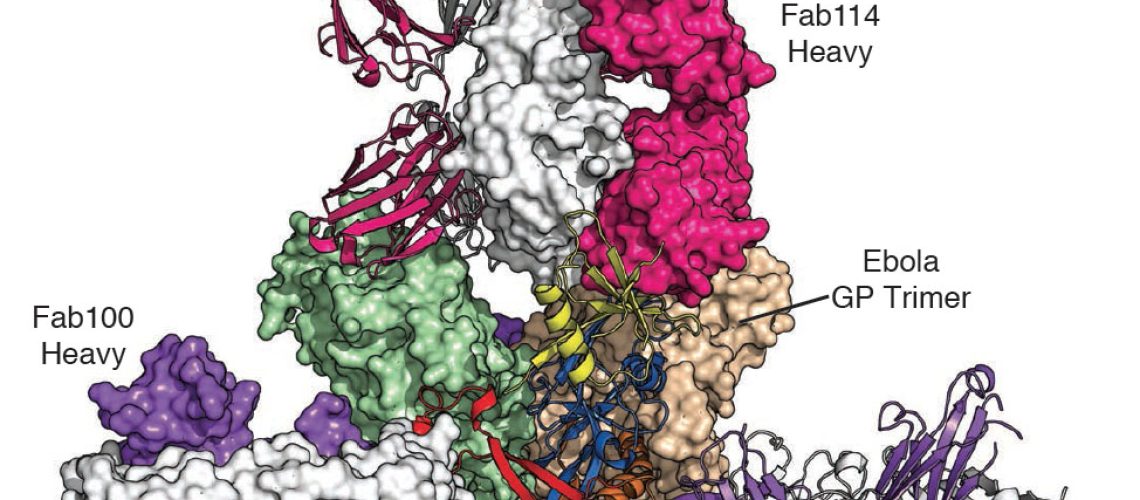
The new drug is based on the monoclonal antibody mAb114, which was isolated and characterised in the laboratory of Antonio Lanzavecchia at the Institute for Research in Biomedicine (IRB, affiliated to Università della Svizzera italiana) in collaboration with Humabs BioMed SA (subsidiary of Vir Biotechnology Inc. of San Francisco) and NIH (U.S. National Institute of Health). The results of the work carried out in Bellinzona, in addition to being published within two different articles in the prestigious scientific journal Science in 2016 and then in The Lancet in January 2019, contributed to the successful outcome of clinical field trials coordinated by the NIH in the Democratic Republic of Congo, where a new outbreak of the deadly virus broke out in 2018. This success was also possible thanks to the important support, over the years, from the DARPA (U.S. Defense Advanced Research Projects Agency). This is the first Ebola monotherapy (a single antibody-based treatment) to be approved.