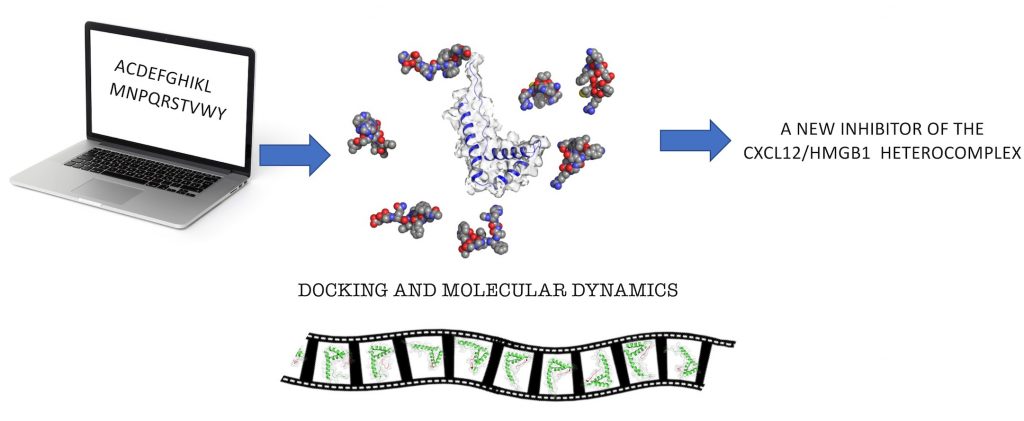
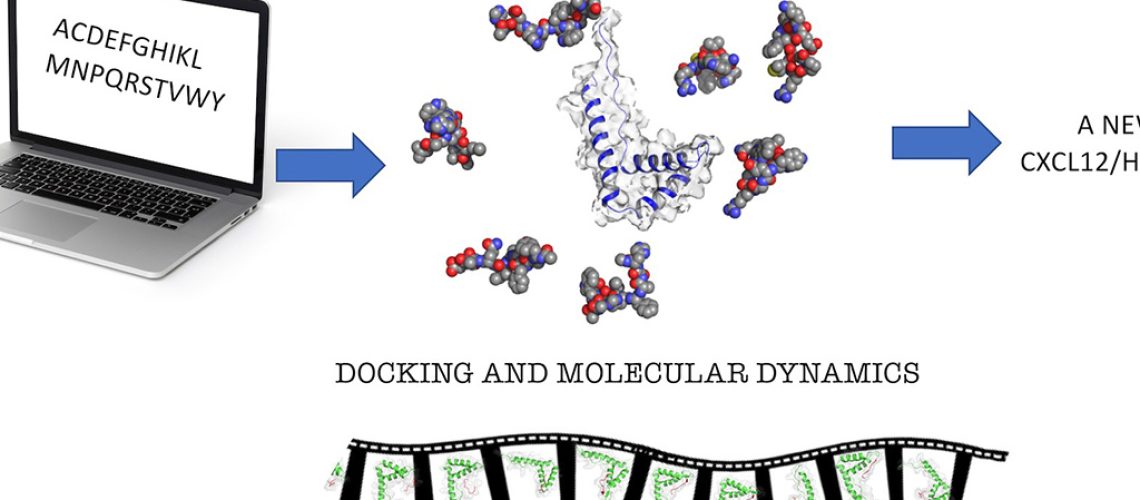
During inflammatory reactions, the production and release of chemotactic factors guide the recruitment of selective leukocyte subpopulations. The chemokine CXCL12 and the alarmin HMGB1, both released in the microenvironment, can form a heterocomplex, which exclusively acts on the chemokine receptor CXCR4, enhancing cell migration and, in some pathological conditions such as Rheumatoid Arthritis exacerbates the immune response.
A study performed by the Cavalli and Uguccioni labs and published in the Journal of Medicinal Chemistry, reports about the identification and characterization of the first peptide inhibitor of the CXCL12/HMGB1 heterocomplex.
In the study, computational methods have been used to identify a potent and selective ligand for HMGB1 (HBP08) at the binding site with CXCL12. Biophysical, structural biology techniques, and functional assays were used to fully characterize the effect of HBP08 on leukocyte migration, to confirm the inhibition of the activity of the heterocomplex.
This study represents an important step towards an effective pharmacological modulation of the CXCL12/HMGB1 heterocomplex in inflammatory and autoimmune diseases.
Article
Systematic Development of Peptide Inhibitors Targeting the CXCL12/HMGB1 Interaction.
Sgrignani, J., V. Cecchinato, E. M. A. Fassi, G. D’Agostino, M. Garofalo, G. Danelon, M. Pedotti, L. Simonelli, L. Varani, G. Grazioso, M. Uguccioni and A. Cavalli
J Med Chem. 2021, DOI: 10.1021/acs.jmedchem.1c00852.