on March 7, 2019
A collaborative project between the Institute for Research in Biomedicine (IRB) affiliated to the Università della Svizzera italiana (USI), the Institute for Protein Design, Washington University, USA, the Center for Molecular Medicine, Karolinska Institutet, Stockholm, Sweden and the Fred Hutchinson Cancer Research Center, Seattle, USA provides new hope for the development of an efficient vaccine against Respiratory Syncytial Virus (RSV). This study was published today in the renowned scientific journal Cell.
Background
Human Respiratory syncytial virus (RSV) is an enveloped RNA virus and a worldwide public health concern. Infection of healthy adults by RSV typically results in mild respiratory symptoms. However, the disease can be very serious in infants and elderly. RSV infection is the second cause of infant mortality worldwide after malaria and accounts for a substantial hospitalization burden in both age groups in developed countries. The virus displays on its envelope three proteins (F, G, and SH). F is the fusion protein responsible for merging the viral membrane with the human host cellular membranes. Previous studies identified that F-specific antibodies account for the majority of neutralizing activity in the sera of infected humans and F is therefore the focus of many vaccine efforts. Nevertheless and despite substantial efforts, an effective vaccine for RSV has not yet been developed. This can be explained by the nature of viral fusion proteins, which undergo major structural rearrangements as they transitions from a prefusion to a postfusion state upon host infection. Subunit vaccine candidates were until recently limited to the more stable postfusion state, but in clinical trials, only modest increases in neutralizing antibodies were observed.
The discovery
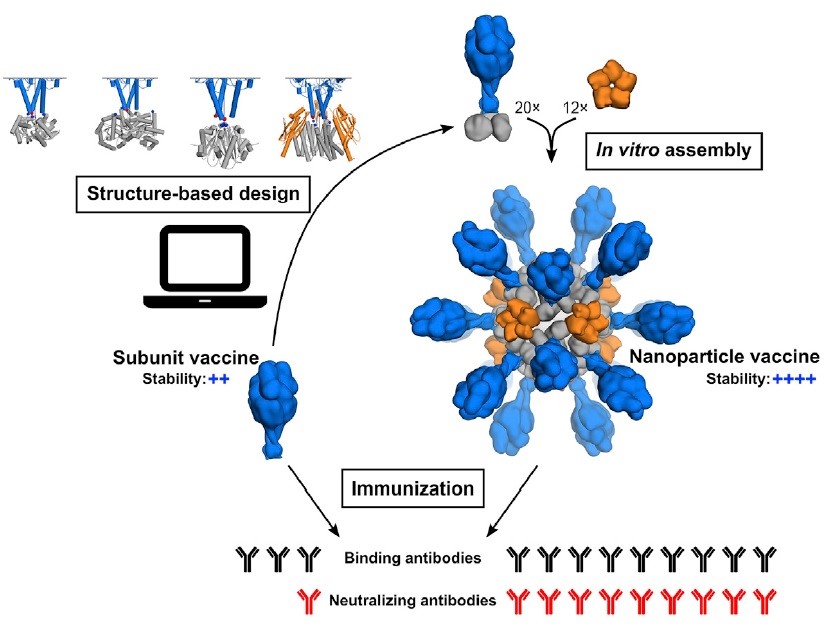
Our study describes the structure-based design of a self-assembling protein nanoparticle presenting a stabilized variant of the fusion glycoprotein trimer used by human RSV to infect individuals. This vaccine candidate induced potent neutralizing antibody responses, which are approximately ten-fold higher than the one elicited by the vaccines currently in clinical trials.
 |
|
A computationally designed self-assembling nanoparticle that displays 20 copies of a trimeric viral protein induces potent neutralizing antibody responses. Structural model of F–RSV nanoparticle and schematic representation of its in vitro assembly process. Each nanoparticle presents the antigen on its surface as a dense repetitive array. |
Our study provides new hope for the development of an efficient vaccine against RSV and prove that nanoparticle based design is a viable option to develop vaccines for other infective diseases. This is particularly relevant in cases where the target antigen is unstable or poorly immunogenic. The study was jointly coordinated by Laurent Perez at the Institute for Research in Biomedicine in Bellinzona (IRB, affiliated to USI) and Neil King at the Institute for Protein Design, Department of Biochemistry, Washington University, USA. The study also involved the Center for Molecular Medicine, Karolinska Institutet, Stockholm, Sweden and the Fred Hutchinson Cancer Research Center, Seattle, USA. The work was supported by the Bill and Melinda Gates Foundation.
The importance of this study is witnessed by Rino Rappuoli, a world famous immunologist currently serving as Chief Scientist and Head of External R&D at GlaxoSmithKline (GSK) Vaccines, who wrote a comment on the perspectives of our study within the same Cell issue.
Article:
Jessica Marcandalli, Brooke Fiala, Sebastian Ols, Michela Perotti, Willem de van der Schueren, Joost Snijder, Edgar Hodge, Mark Benhaim, Rashmi Ravichandran, Lauren Carter, Will Sheffler, Livia Brunner, Maria Lawrenz, Patrice Dubois, Antonio Lanzavecchia, Federica Sallusto, Kelly K. Lee, David Veesler, Colin E. Correnti, Lance J. Stewart, David Baker, Karin Loré, Laurent Perez, Neil P. King. Cell.
J. Marcandalli and B. Fiala contributed equally to this work. L. Perez and N. P. King contributed equally to this work and are co-corresponding authors.