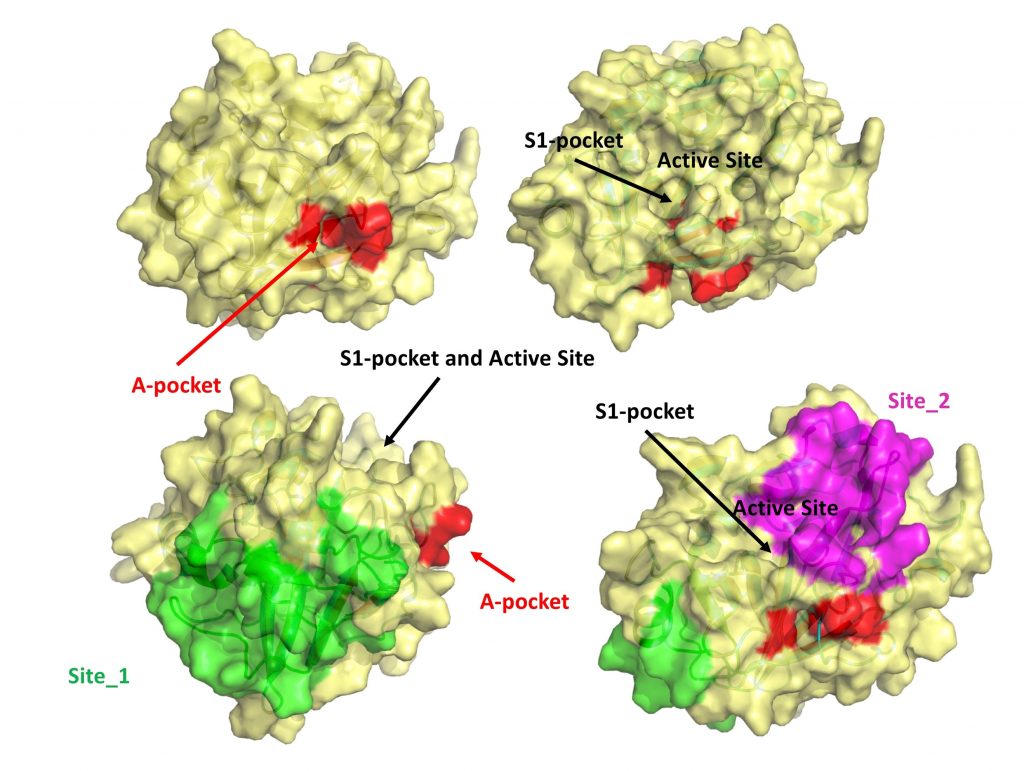
A new paper from the Cavalli’s lab describes the computational identification of a novel putative allosteric binding site in the protein TMPRSS2.
The membrane bound serine protease TMPRSS2 is one of the proteases responsible for the proteolytic priming of SARS-CoV-2 spike protein which leads to the release of the fusion peptide. Its inhibition represents, therefore, an attractive strategy to prevent viral infection.
To identify the allosteric pocket, Sgrignani and co-workers performed several multi-microsecond molecular dynamics simulations of the catalytic domain of TMPRSS2 alone and in complex with an inhibitor (bromhexine). From the analysis of the computer simulations, they were able to show that the binding of small molecules or peptides to this pocket alter the structure of the catalytic site, thus, significantly reducing its ability to bind and process the substrate.
This study, totally carried out at IRB, represents a first step toward the development of more selective and potent antiviral drugs capable of modulating TMPRSS2 activity.
Article
Computational identification of a putative allosteric binding pocket in TMPRSS2.
Sgrignani and A. Cavalli.
Front. Mol. Biosci. 2021. DOI: 10.3389/fmolb.2021.666626